Sichuan Techairs Co., Ltd |
|
Verified Suppliers
|
|
Chemical Fiber PTA Refined SMR Hydrogen Plant 330 M3/H Mature Process Technology
Chemical Fiber PTA Refined 330 m3/h SMR Hydrogen Plant
Technology Application:
All kinds of hydrogen-rich industrial exhausts (such as chlor-alkali exhaust, coke oven gas, synthetic ammonia tail gas, by-product gas from refining, released gas from methanol, PTA exhaust etc.) are separated and purified for high purity hydrogen through the pressure swing adsorption process, and the technology has reached the world advanced level.
Technical indexes
Purity:99~99.999%
Capacity:5~100000Nm3/h
Yield:≥90%
Features and advantages
Mature process technology and low costs of hydrogen production.
Wide source of raw material and high purity of product gas.
The Two-high THiPSATM technology is applied.
The special adsorbent has a long service life and low operating costs.
Special programmable control valve is of high automatic degree.
Process Schematic
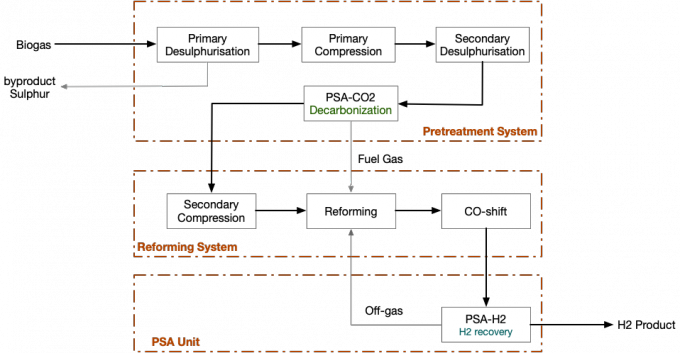
Advantages
Variable degrees of customization versus standardization to meet project requirements
Focus on best in class plant availability and reliability benefiting from Air Liquide’s operational experience
Dedicated standard plant program for small hydrogen capacities with high degree of modularization allowing for execution time of less than 15 months Free On Board (FOB)
Natural gas will be mixed with process steam as per the proportion 3.2~3.8 after desulfurized to 0.1PPm and below and then flow into reforming pipe after entering mixed gas preheating coil pipe and being further preheated to 530~580℃. In catalyst bed, H2 and CO will be generated via reaction of methane and steam, and CO will be generated into CO2 after reacting with steam. Heat required for methane reforming is provided by burning mixed fuel gas. The main reaction of nickel catalyst is shown below:
CH4+H20(steam) = CO +3H2-49200Kcal/Kmol(reforming reaction)
CO+H20(steam) = CO2 +H2+9840Kcal/Kmol (shift reaction)
Cracking reaction of higher alkane (400~600℃)
CnH2n+2+nH2O(steam) =(2n+1) H2 + n CO
