| Description | Lithium carbonate (molecular structure is Li2CO3, English name is
lithium carbonate) as a colorless monoclinic crystal or white
powder. Density is 2.11. Melting point is 618 ℃. Without
deliquescence, it is stable in the air. Low solubility in water,
the solubility decreases with increasing temperature. Solubility in
cold water is greater than hot water. It is Soluble in dilute acid,
insoluble in alcohol and . Carbon dioxide is introduced into the
aqueous suspension of lithium carbonate, lithium carbonate is
converted to lithium acid carbonate and dissolved. If the solution
of lithium acid carbonate is heated and then it releases carbon
dioxide and precipitates lithium carbonate. The nature of the
lithium carbonate may be used to remove impurities from lithium
carbonate. Since lithium ion has a strong polarizability, thus
thermal stability of lithium carbonate is worse than other alkali
metal carbonate, when heated to above the melting point, it will
decompose to produce carbon dioxide and lithium oxide.
 |
| Chemical Properties | Lithium carbonate is a white monoclinic crystalline solid.
Typically for carbonates, lithium carbonate reacts with acids
stronger than carbon dioxide or carbonic acid to yield the lithium
salt of the acid and carbon dioxide. The reactions may be carried
out in a solution, as an aqueous slurry, or, less effectively, with
solid lithium carbonate.
Lithium carbonate exhibits a low water solubility for an alkali
metal carbonate. The solubility decreases with increasing
temperature. It is not hygroscopic and is generally stable when
exposed to the atmosphere. In fact, it is the normal end compound
encountered when many basic lithium compounds are exposed to the
atmosphere. Lithium carbonate may be dissolved in water by
conversion to the hydrogen carbonate. Releasing carbon dioxide by
heating the solution of lithium hydrogen carbonate causes
reprecipitation of the lithium carbonate. |
| Uses | The lithium carbonate industry is a global high monopoly industry,
the current production capacity is mainly concentrated in three
foreign manufacturers of SQM, FMC, Chemetall and so on.
Industrial lithium carbonate is used in the manufacture of other
lithium salts, such as lithium chloride and lithium bromide and so
on. It also acts as lithium oxide materials in enamel, glass,
pottery and porcelain enamel, and it is also added to the
electrolytic cell for electrolysis of aluminum to increase the
current efficiency and reduce the internal resistance of the cell
and the bath temperature. In medicine, it is mainly used for the
treatment of mania, can improve their emotional disorders for
schizophrenia. It has the effect of elevating peripheral
leukocytes; can be used for synthetic rubber, dyes, semiconductor
and military defense industry and so on; for the production of
lithium tantalate, lithium niobate and other acoustic grade single
crystal, optical grade monocrystalline etc; for preparation of the
acoustic grade single crystal.
Battery grade lithium carbonate is mainly used for the preparation
of lithium cobalt oxide, lithium manganese oxide, ternary
materials, lithium iron phosphate and other lithium ion battery
cathode materials; used in a matrix modifier; as aneuroprotective
effect of lithium carbonate in amyotrophic lateral sclerosis. |
| Toxicity | Lithium carbonate has a significant stimulating effect, firstly has
damage on the gastrointestinal tract, kidney and central nervous
system. Toxicity order of lithium compounds is Li <LiCl
<Li2CO3, maximum allowable concentration: Lithium condensation
and fragmentation aerosol were 0.05 mg/m3 and 0.5 mg/m3.
Wear rubber gloves and protective masks when working, in order to
protect the respiratory organs against dust. |
| Preparation | Lithium carbonate is obtained as an intermediate product in
recovery of lithium metal from its ore, spodumene (See Lithium). It
is prepared by mixing a hot and concentrated solution of sodium
carbonate with lithium chloride or sulfate solution.
Li2SO4+ Na2CO3→Li2CO3+ Na2SO4 |
| Reactions | Lithium carbonate reacts with dilute acids, liberating carbon
dioxide:
Li2CO3+ HCl →LiCl + CO2+ H2O
Thermal decompostion yields lithium oxide and carbon dioxide:
Li2CO3 → Li2O + CO2
Reaction with lime produces lithium hydroxide:
Li2CO3+ Ca(OH)2→2LiOH + CaCO3
The carbonate reacts with molten aluminum fluoride converting to
lithium fluoride:
3Li2CO3+ 2AlF3 → 6LiF + 3CO2+ Al2O3
It combines with carbon dioxide in aqueous slurry forming soluble
bicarbonate, which decomposes to carbonate upon heating:
Li2CO3+ CO2+ H2O →2LiHCO3
The bicarbonate can not be separated in solid form. It exists only
in solution when carbonate dissolves in water saturated with
CO2under pressure. |
| Chemical Properties | Lithium carbonate is a white hygroscopic powder. |
| Physical properties | White monoclinic crystals; refractive index 1.428; density 2.11
g/cm3; melts at 723°C; decomposes at 1,310°C; low solubility in water
(1.54 g/100g) at 0°C; 1.32 g//100g at 20°C), solubility decrease
with temperature (0.72g/100g at 100°C); insoluble in and ethanol. |
| Uses | The most common lithium drug is lithium carbonate, which possesses
antimania action. It is presumed that lithium alters the transport
of sodium ions in neurons, thus influencing the intercellular
contents of catecholamines, normalizing the mental state and not
causing general lethargy. It is used for mania conditions of
various origins, preventative measures, and for treating affective
psychoses. |
| Uses | Lithium carbonate is used as a compound for producing metallic
lithium. Lithium carbonate is the result of treating the mineral
spodumene with sulfuric acid and then adding calcium carbonate. It
is used as an antidepressant. |
| Uses | In the production of glazes on ceramic and electrical porcelain. |
| Preparation | Lithium carbonate is prepared by the precipitation of lithium ion
by carbonate ion from an aqueous solution. Still another process,
which is carried out on a smaller scale, is the reaction of a
solution of lithium hydroxide with carbon dioxide gas. Lithium
carbonate precipitates and is recovered from the supernatant
solution. |
| Definition | lithium carbonate: A white solid,Li2CO3; r.d. 2.11; m.p. 723°C;
decomposesabove 1310°C. It is producedcommercially by treating the
ore with sulphuric acid at 250°C andleaching the product to give a
solutionof lithium sulphate. The carbonateis then obtained by
precipitationwith sodium carbonate solution.Lithium carbonate is
used in the preventionand treatment of manicdepressivedisorders. It
is also usedindustrially in ceramic glazes. |
| Indications | Lithium inhibits thyroidal incorporation of I- into Tg, as well as the secretion of thyroid hormones, but it does
not inhibit the activity of the Na+-I- symporter or the accumulation of I- within the thyroid. Lithium offers no particular advantage over
drugs of the thionamide class but may be employed for temporary
control of thyrotoxicosis in patients who are allergic to both
thionamides and iodide. |
| Brand name | Eskalith (GlaxoSmithKline); Lithane (Bayer); Lithobid (JDS);
Lithonate (Solvay Pharmaceuticals). |
| General Description | Lithiumcarbonate (Eskalith, Lithane) and lithium
citrate(Cibalith-S) are the salts commercially available in
theUnited States. |
| Reactivity Profile | A base. Decomposed by acids with the evolution of carbon dioxide.
Fluorine burns fiercely on contact with Lithium carbonate. |
| Safety Profile | Human carcinogenic data. Poison by intraperitoneal and intravenous
routes. Moderately toxic by ingestion and subcutaneous routes.
Human systemic effects by ingestion: toxic psychosis, tremors,
changes in fluid intake, muscle weakness, increased urine volume,
nausea or vomiting, allergic dermatitis. Human reproductive effects
by ingestion: effects on newborn, including Apgar score changes and
other neonatal measures or effects. Human teratogenic effects by
ingestion: developmental abnormalities of the cardiovascular
system, central nervous system, musculoskeletal and
gastrointestinal systems. An experimental teratogen. Experimental
reproductive effects. Experimental carcinogen producing leukemia
and thyroid tumors. Human mutation data reported. Used in the
treatment of manic-depressive psychoses. Incompatible with
fluorine. See also LITHIUM COMPOUNDS. |
| Chemical Synthesis | Lithium carbonate is synthesized by reacting lithium salts with
soda or potash, followed by purification of the salt, which is not
readily soluble [75]. 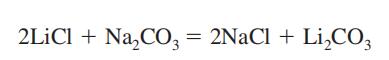
|
| Potential Exposure | Lithium carbonate is used in treatment of manic-depressive
psychoses; to make ceramics and porcelain glaze; varnishes, dyes,
pharmaceuticals, coating of arc-welding electrodes; battery alloys;
nucleonics, luminescent paints; glass ceramics; lubricating
greases; in aluminum production |
| Shipping | UN2811 Toxic solids, organic, n.o.s., Hazard Class: 6.1; Labels:
6.1-Poisonous materials, Technical Name Required |
| Purification Methods | Crystallise it from water. Its solubility decreases as the
temperature is raised. The solubility in H2O is 1.3% at ~10o, and
0.7% at ~100o. [D.nges in Handbook of Preparative Inorganic
Chemistry (Ed. Brauer) Academic Press Vol I p 987 1963, Caley &
Elving Inorg Synth I 1 1939.] |
| Incompatibilities | The aqueous solution is a strong base. Reacts violently with acids,
powdered calcium and fluorine.Incompatible with oxidizers
(chlorates, nitrates, peroxides, permanganates, perchlorates,
chlorine, bromine, fluorine, etc.); contact may cause fires or
explosions. Keep away from alkaline materials, strong acids,
powdered calcium, fluorine, moisture. Corrodes aluminum, copper,
zinc. |