| Description | Lanosterol occurs naturally in the eyes of mammals and has been
shown to dissolve cataracts by revitalizing damaged
cataract-causing crystallin proteins. Lanosterol’s amphipathic
nature is believed to play a large role in its ability to prevent
and reverse cataract formation.  |
| Source | Lanosterol is the first sterol in lipid biosynthetic pathway, which
is initially converted by acetyl-CoA. The complex process of
lanosterol synthesis involves several enzymes, including
3-hydroxy-3-methylglutaryl-coenzyme A (HMG-CoA) reductase, squalene
epoxidase, and lanosterol synthase (LSS). LSS is a microsomal
enzyme that functions as a downstream element in the lanosterol
biosynthetic pathway, catalyzing the cyclization of the linear
2,3-monoepoxysqualene to lanosterol. |
| Function | A cytochrome P450 monooxygenase involved in sterol biosynthesis.
Catalyzes 14-alpha demethylation of lanosterol and
24,25-dihydrolanosterol likely through sequential oxidative
conversion of 14-alpha methyl group to hydroxymethyl, then to
carboxylaldehyde, followed by the formation of the delta 14,15
double bond in the sterol core and concomitant release of formic
acid (PubMed:20149798, PubMed:8619637). Mechanistically, uses
molecular oxygen inserting one oxygen atom into a substrate, and
reducing the second into a water molecule, with two electrons
provided by NADPH via cytochrome P450 reductase. |
| Biosynthesis pathway | The lanosterol pathway refers to a segment of the cholesterol
biosynthesis pathway comprising twelve enzymes, namely acetyl-CoA
acetyltransferase, hydroxymethylglutaryl-CoA synthase,
hydroxymethylglutaryl-CoA reductase, mevalonate kinase,
phosphomevalonate kinase, diphoshomevalonate decarboxylase,
isopentenyl-diphosphate delta isomerase, geranylgeranyl diphosphate
synthase, farnesyl diphosphate synthase, squalene synthase,
squalene monooxygenase and lanosterol synthase. The lanosterol
pathway describes the stages of cholesterol biosynthesis between
the conversion of substrates acetyl CoA and acetoacetyl CoA to
(S)-3-hydroxy-3-methylglutaryl-CoA, through to the formation of
intermediate metabolite lanosterol, the precursor of cholesterol.
Metabolites of the lanosterol pathway are either directed to the
synthesis of cholesterol and other sterols, or to side branches of
the pathway through which they are converted to isoprenoids and
other non-sterols. |
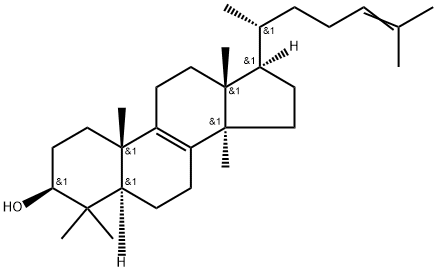
| Definition | ChEBI: A tetracyclic triterpenoid that is lanosta-8,24-diene
substituted by a beta-hydroxy group at the 3beta position. It is
the compound from which all are derived. |
| Purification Methods | If very impure, then it should be acetylated, converted to the
dibromide acetate [crystallised from EtOAc with slow cooling, m
168-170o, [] D +214o (CHCl3)], de-brominated with Zn dust to give
the acetate (below) which is recrystallised from 3-4 parts of
Me2CO/MeOH (4:1) and hydrolysed as for stigmasterol (below).
Recrystallise it from anhydrous MeOH. Dry it in vacuo over P2O5 for
3hours at 90o. The purity is checked by proton magnetic resonance.
The acetate crystallises from MeOH with m 131-133o and [ ] 25D +62o
(c 1,CHCl3). [Block & Urech Biochemical Preparations 6 32 1958.
van Tamelen et al. J Am Chem Soc 104 6479, 6480 1982, Beilstein 6
III 2880, 6 IV 4188.] |