Rapid Test Kit Antige Self Testing Rapid Test Kit COVID-19 Antigen
Detection Self Test Kit
Intend Use
- It's a qualitative detection
- specimen sample is nasal swab
- The test result can be used for early isolation of patients with
suspected infection
- To be notic, the result cannot be used as only diagnosis basis of
SARS-CoV-2 infection
- Negative results do not rule out SARS-CoV-2 infection and should
not be used as the sole basis for treatment.
- Further nucleic acid detection should be carried out for suspected
population whose antigen test result is positive or negative
Product Details
| Item | Value |
| Model Number | LX-401320 |
| Type | 20 Tests/Kit |
| Warranty | 24 Months |
| Power Source | Instruction Manual |
| Quality Certification | CE,MSDS |
| Safty Standard | ISO13485,ISO9001 |
| Smple Type | Nasal Swab |
| Sample volume | 3 Full drops |
| Test Speed | 10 minutes < x < 15 minutes |
| Total Accuracy | 99.17% |
| Sensitivity | 97,45% |
| Specificity | 100% |

Product Feature
- No pre-processing required, simple operation, do not need any
auxiliary equipment
- Rapid test, easy to interpret the results in 15 minutes
- Transported and stored at room temperature
- High Accuracy, Sensitivity and Specificity


Main Components
- 20 Test Cassettes
- 20 Sample tubes
- 20 Extraction Buffer
- 20 Swabs
- 1 Tube stand
- 1 Package Insert
Use Step
- Open the sealed pouch and remove the test casstette. Lay it face up
on a clean, dry and flat surface.
- Unpack the sample extraction, add all of the sample extraction into
the sample tube and then put the tube into tube stand.
- Gently, insert the entire absorbent tip of the swab (around 1.5 cm)
into your nostril. Rotate walls of your nostril 5 times or more.
Use the same swab to repeat steps in the other nostril.
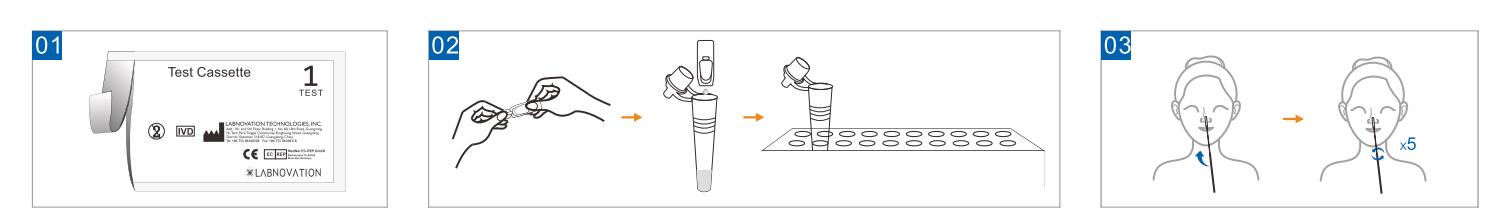
- Insert the swab into the ssample tube with extraction buffer. Mix
well. Mix well and squeeze the swab 10-15 times by compressing the
walls of the tube against the swab. Roll the swab head against the
inner wall of the tubes as you remove it.
- Close the cape of the sample tube. Add 3 full drops of the mixed
solution vertically into the sample well(S) of the test cassette.
- Read the result 15-20minutes after adding the sample. Result got
after 20 minutes is invalid.
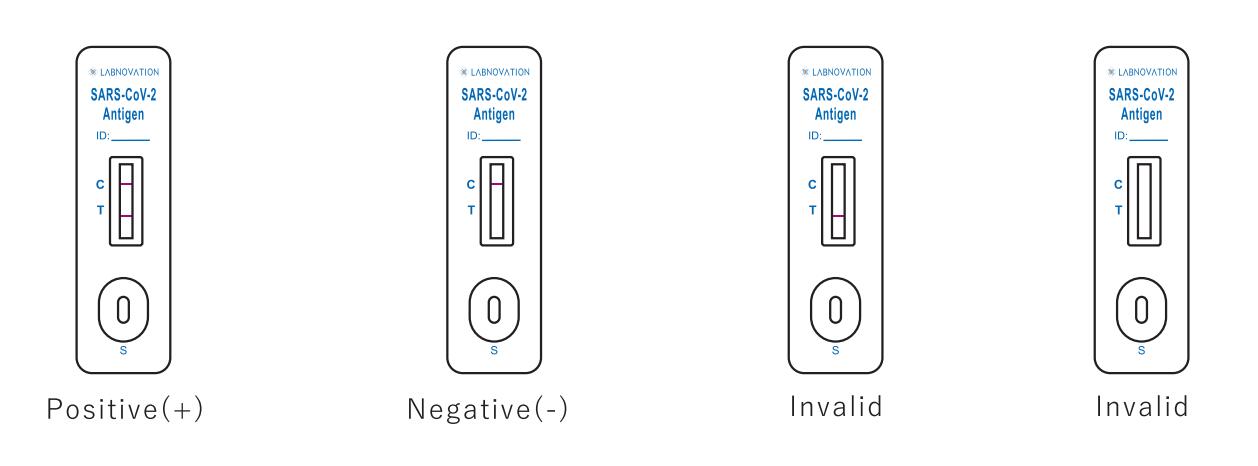
Result Interpretation
POSITIVE: Two colored bands appear on the membrane. One band
appears in the control region (C) and another band appears in the test region (T).
NEGATIVE: Only one colored band appears, in the control region (C). No apparent colored band appears in the test region (T).
INVALID: If there is no Control line (C) or only a Test line (T) in the result window, the test did not run correctly and the
results are not valid.
Virus Sources
| Global high frequency mutation | Alpha / B.1.1.7(U.K.) | Beta I B.1.351(South Africa) |
| Gemma I P.1(Brazil) | Kappa I B.1.617.1(India) | Delta I B.1.617.2(India) |
| C.37,ect | Alpha I B.1.17(U.K.) | B.1.36.16.etc |
| A.2.5,etc | A.23.1 | Alpha I B.1.17(U.K.) |
| B.1.1.33.etc | C.1.1.etc. | etc. |
FAQ
- MOQ: MOQ limit that is 10000 pieces.
- Delivery Date: After order confirmed, we will arrange your order
immediately and offer you an estimated delivery date.
- Shipment: We choose Air cargo or Ocean cargo
- Is it possible to use our own packing box/specified packing
box: For bulk order, the box can be customized, we will confirm design
before packaging.
- How to get a quotation: Please tell us the products and quantity you need, we will offer
you our best price.
- Can you design for us: We accept various OEM/ ODM orders.
Other Information
- This kit is a qualitative detection, which cannot determine the
exact content of antigen.
- The test is intended for use outside the body only.
- Not to be taken internally. Avoid sample buffer contact with skin
and eyes.
Keep out of the reach of children. Any child under age 18 shouldn’t
perform the test without parental guidance, or professional aid.
Not following the exact instructions can affect the outcome of the
test. The final diagnosis must be confirmed by a physician.
Do not use the test if the packaging is damaged. Do not use broken
test components.
Certificate







