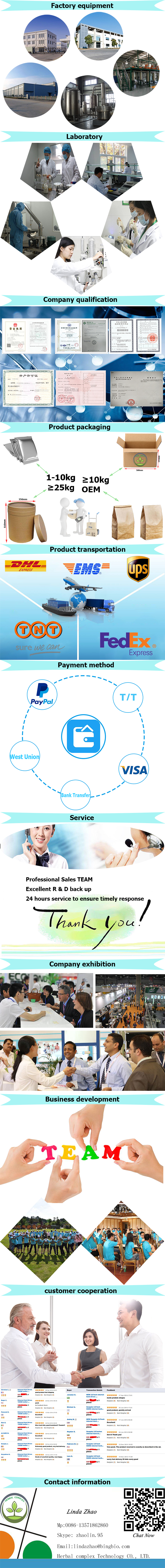
Complex Nature Biotechnology CO., LTD. |
Herbal Medicine Dodder Extract For Eyesight Capsule
Product Details
Product Name: China Dodder Extract
Latin Name: Cuscuta chinensis Lam.
Appearance: Light Yellow powder
Part of the Plant Used:Root/seed
Extract Method: Grain Alcohol/Water
Product Specification : 10:1,20:1


Cistanche, also known as essence, is the current world to the brink of extinction of species, highmedicinal value, known as "desert ginseng" reputation, is found more than 60 kinds of Chinese Traditional Medicine in the highest grade drugs, contains large amounts of amino acidshave a
great deal of benefit, cystine, vitamins and minerals rare nutrition nutritious ingredients, male
kidney, testicles, penis, cavernous and other sexual organs, impotence, premature ejaculation is more immediate, and fulfilled as a god.
Product Analysis
| Item | Specification | Result | Method |
| BasicProduct Information | |||
| Product name | dodder extract | ||
| Part of the Plant | seed | Conform | / |
| Country of Origin | China | Conform | / |
| Organoleptic Data | |||
| Appearance | Fine powder | Conform | NLS-QCS-1008 |
| Color | Light Yellow powder | Conform | GB/T 5492-2008 |
| Odor& Taste | Characteristic | Conform | GB/T 5492-2008 |
| Process Data | |||
| Solvent(s) Used | Water and ethyl alcohol | Conform | / |
| Extract ration | / | Conform | |
| Content | Paeoniflorin≥98% | 98.13 | |
| Drying Method | Spray drying | Conform | / |
| Excipient | Maltodextrin | Conform | / |
| Physical Characteristics | |||
| Particle Size (80 mesh) | 98.0%pass 80mesh | Conform | GB/T 5507-2008 |
| Moisture | <5.0% | 2.5% | GB/T 14769-1993 |
| Ash Content | <4.0% | 1.2% | AOAC 942.05, 18th |
| Solvent Residue | Eur. Pharm | Conform | NLS-QCS-1007 |
| Heavy Metals | |||
| Total Heavy Metals | <10 ppm | Conform | USP <231>, method II |
| Arsenic | <1.0 ppm | Conform | AOAC 986.15, 18th |
| Lead | <1.0 ppm | Conform | AOAC 986.15, 18th |
| Mercury | <0.5 ppm | Conform | AOAC 971.21, 18th |
| Pesticide Residue | |||
| 666 | <0.2ppm | Conform | GB/T5009.19-1996 |
| DDT | <0.2ppm | Conform | GB/T5009.19-1996 |
| Microbiology | |||
| Total Plate Count | <1,000cfu/g | 13cfu/g | AOAC 990.12, 18th |
| Total Yeast & Mold | <100cfu/g | 3cfu/g | FDA (BAM) Chapter 18, 8th Ed. |
| E. Coli | Negative | Negative | AOAC 997.11, 18th |
| Salmonella | Negative | Negative | FDA (BAM) Chapter 5, 8th Ed. |