Rapid Antibody Test Kit COVID-19 Neutralizing Antibody Rapid test
Kit 20 Tests For Professional Use
Intended Use
- The SARS-COV-2 Neutralizing Antibody Rapid Test Kit
is intended for the semi-quantitative determination of
neutralizing antibodies (NAbs) to SARS-COV-2 that block the
interaction between the receptor binding domain (RBD) of the viral
spike glycoprotein with the ACE2 cell surface receptor in human
serum, plasma (EDTA and sodium citrate), whole blood specimens.
- The test is intended use as an aid in identifying individuals with
an adaptive immune response to SARS-COV-2, indicating recent or
prior infection or assisting in evaluating the effectiveness of the
vaccine clinical trials and mass vaccination.
- Results are for the detection of total neutralizing antibodies to
SARS-COV-2.
Specifications
| Test Item | Neutralizing Antibody Test Kit |
| Number | LX-401701 |
| Sample Volume | Whole blood , Serum, Plasma |
| Package | 20 Tests/Kit |
| Specificity | 98.00% |
| Sensitivity | 97.67% |
| Accuracy | >95% |
| nspection Method | Immunochromatography |
| Shelf Life | 18 Months |
| Type | IVD Antibody Rapid Test |
| Humidity | 35%-60% |
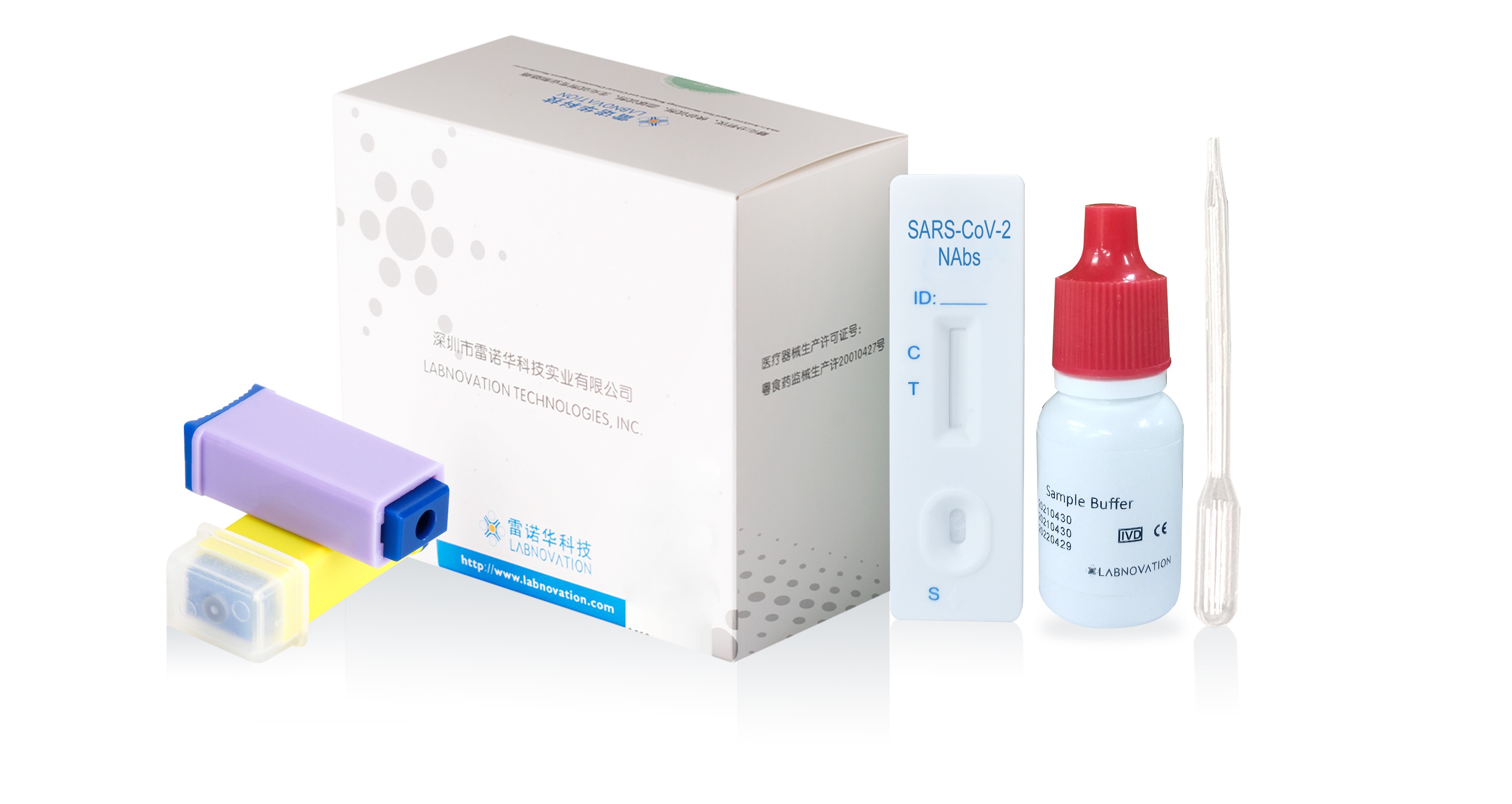
Main Components
- 20 Test cassettes
- 20 Disposable pipettes
- 20 Sterile Safety Lancets
- 1 Sample Buffer
- 1 Colorimetric chart
- 1 Package Insert
Product Feature
- Accurate: the product has a high accurate serology test for
Neutralizing Antibodies detection
- Easy to administer and read results: no need for instrument or
other equipment during the test
- Fast detection: the result appears in 15 minutes, can be used for
testing on a massive scale.
- No specific storage requirement: room temperature (2-30ºC)

Advantage
- Blood testing, Fingerstick whole blood is workable
- Easy to operate, no additional material required to run the assay
- Little Specimen is required.
Principle
The test employs the sandwich immunoassay to detect the total
neutralizing antibodies. The color intensity of the T line is
correlated with the level of the neutralizing Antibody in specimen.
To determine the level of the neutralizing antibody in specimen,
comparing the intensity of test line to the attached colorimetric
chart.
Use Step
- Place the lanceton the test site.
- Push down the lancet.
- Collect the bloodby transfer pipette.
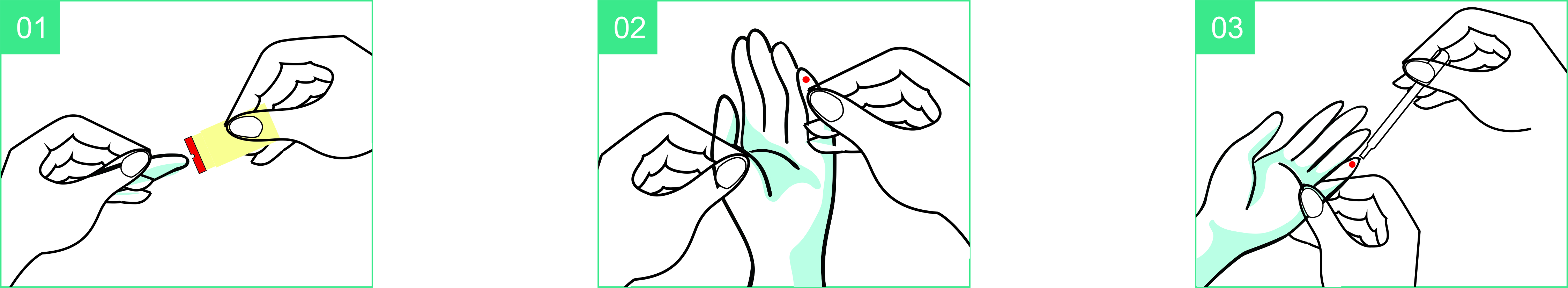
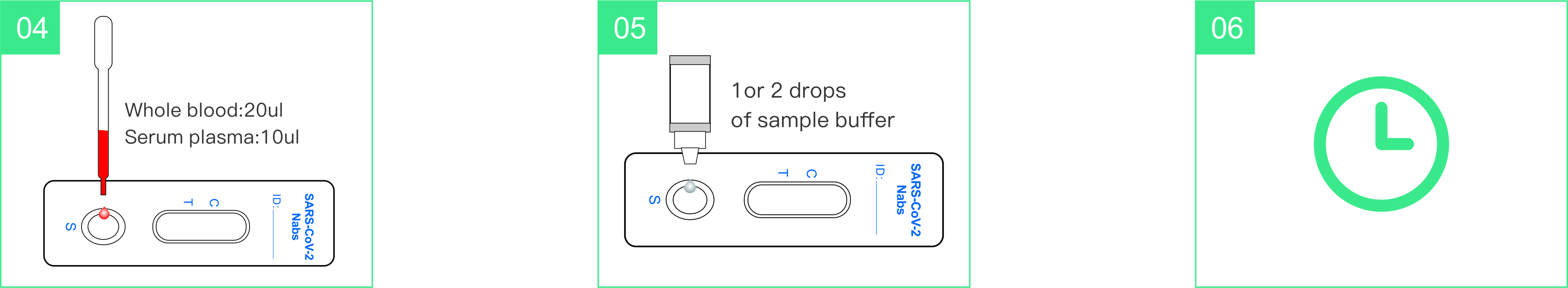
- Add one drop of blood into the sample well.
- Add 1-2 drops (30-60µL) of sample buffer into the sample well
innediately.
- Wait 15 minutes then read the result.
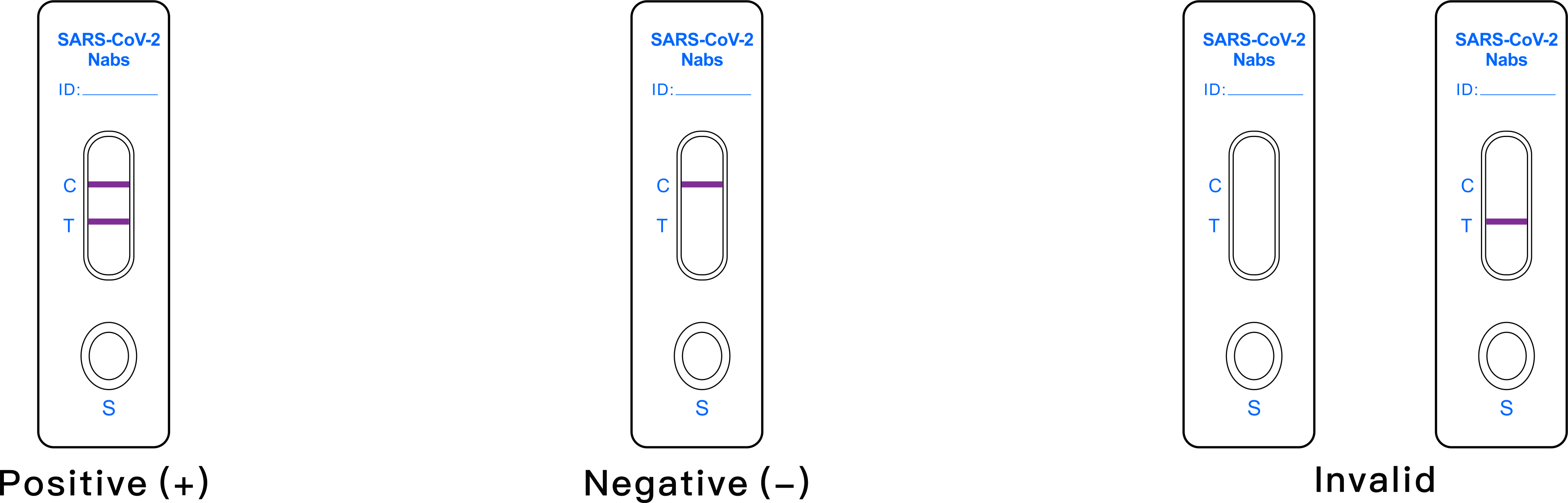
Interpretation of results
- POSITIVE: The T line and C line are both displayed within the
reaction window, indicates the neutralizing antibodies to
SARS-COV-2 are detected. The result is positive.
- NEGATIVE:The C line is displayed within the reaction window only,
indicates absence of neutralizing antibody to SARS-COV-2.The result
is negative..
- INVALID:1. If the control (C) line is not displayed in 15 min, regardless
of whether T line is present, the test result is invalid. It is
recommended that the specimen should be re-tested.
The test result is invalid after 20 min.
FAQ
We have the MOQ limit,which is 10000 pieces.
After order confirmed,we will arrange your order immediately,and
offer you an
estimated delivery date.
Business to business account.
We choose Air cargo or Ocean cargo.
- Trade Company or Manufacturer:
Manufacturer




