Rapid Tets Kit Antigen Rapid Test SARS-CoV-2 Antigen Saliva Rapid
Test Kit
Intend Use
The rapid test kit is intended for the qualitative detection of
SARS-CoV-2 antigen in hunman saliva samples from novel coronavirus
suspected person with symptoms within 9 days from onset.
Product Details
| Item | Value |
| Model Number | LX-401603 |
| Type | 20 Test |
| Warranty | 18 Months |
| Quality Certification | CE, MSDS |
| Safty Standard | ISO13485 |
| Sample volume | 3 Full drops |
| specificity | 99.00% |
| sensitivity | 94.29% |
| Storage Temperature | 2℃-30℃ |
Product Feature
- Fast: Quick test speed, you can read the result within 15 minutes
- Accurate: High accuracy, specificity and sensitivity, accuracy rate
more than 95%
- Protable: Easy to collect sample, you can do the test
anywhere, anytime
- Simple: Easy to operate, one step testing process
Test Principle
- Immunochromatography assay.
- Gold immunochromatographic test principle, double antibody sandwich
method was used to detect SARS-CoV-2 nucleocapsid antigen in the
samples.
- There is virus antigen presence in the sample, the antigen binds
with the corresponding colloidal gold monoclonal antibody and the
coated monoclonal antibody at the detection line to form a compound
and then condenses into a red band, indicating a positive result.
- There is no antigen in the sample, complex cannot be formed at the
detection line, and no red band is shown, indicating negative
result.
- Notic: Whether the sample contains antigen or not, the gold
monoclonal antibody will bind to the enveloped antibody at the
quality control line, form a compound and condense into a red band.
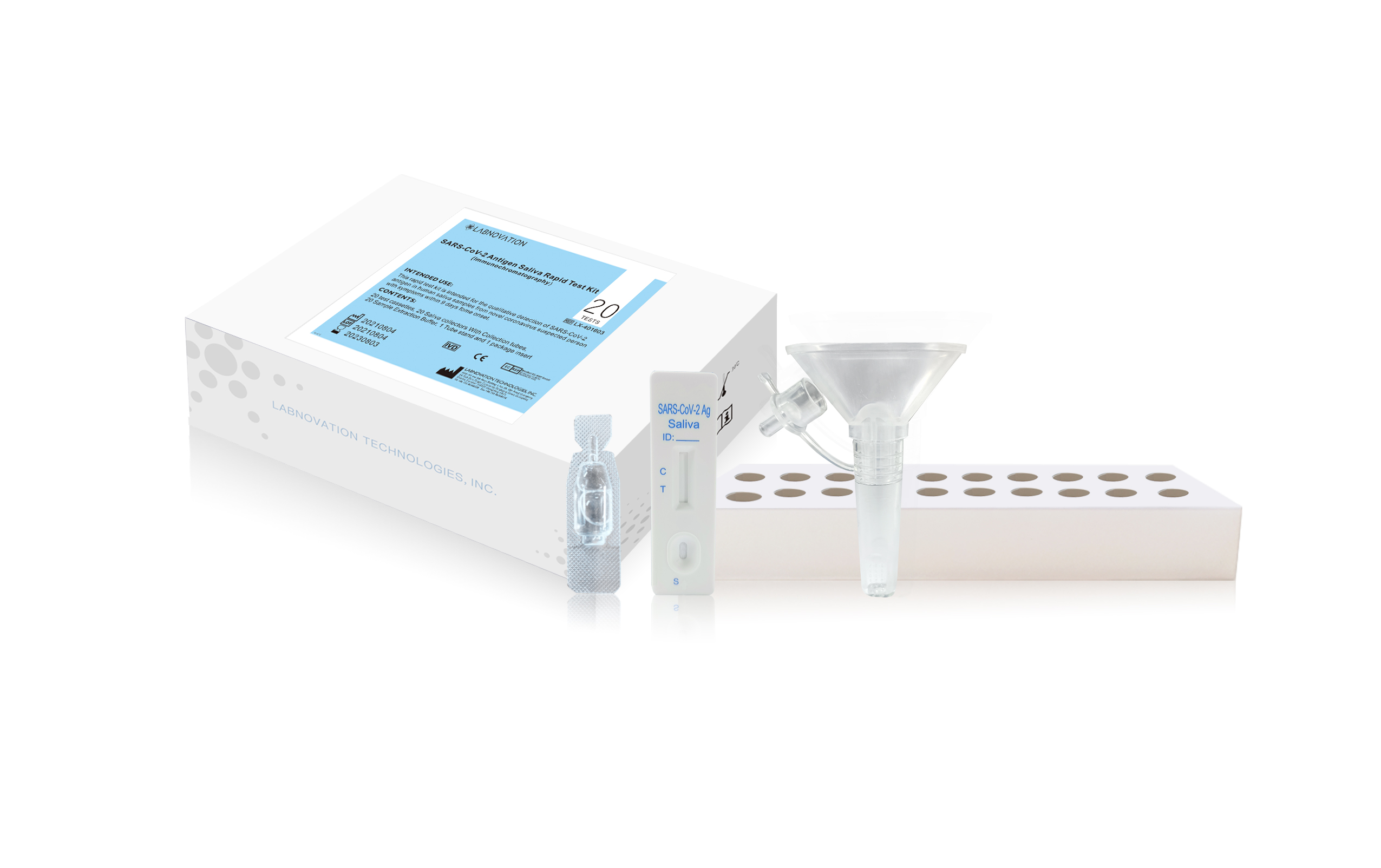
- Main Components
- Test Cassettes
- Saliva Collectors With Collection Tubes
- Sample Extraction Buffers
- Tube stand
- Package Insert

Use Step
- Open the cap of the collection tube and istall saliva collector.
- Put the collection tube with saliva collector close to lips and let
the saliva flow into the collection tube. Tje volum of saliva needs
to be half of 0.5 scal mark(0.25).
- Screw the sample extraction buffer carefully.

- Add all sample extration buffer to the collection tube. Discard the
saliva collector, tighten the cap onto the specimen collection
tube.
- Shake the specimen collection tube at least three time vigorously
to mix the saliva and the extraction buffer. Squeeze bottom of the
collection tube ensure the saliva is thoroughly mixed.
- Add 3 full drops of the solution to the sample well(S). Read the
result at 15 minutes. Do not interpret the result after 20 minutes.

Result Interpretation
POSITIVE: Two (2) distinct colored lines appear. One line should be in the
control region (C) and the other line should be in the test region
(T).
NEGATIVE: One (1) colored line appears in the control region(C). No apparent
colored line appears in the test region (T). The negative result
does not indicate the absence of analytes in the sample, it only
indicates the level of tested analytes in the sample is less than
the minimum detection limit.
INVALID: No colored lines appear, or control line fails to appear,
indicating that the operator error or reagent failure. Verify the
test procedure and repeat the test with a new testing device.

Virus Sources
| Global high frequency mutation | Alpha / B.1.1.7(U.K.) | Beta I B.1.351(South Africa) |
| Gemma I P.1(Brazil) | Kappa I B.1.617.1(India) | Delta I B.1.617.2(India) |
| C.37,ect | Alpha I B.1.17(U.K.) | B.1.36.16.etc |
| A.2.5,etc | A.23.1 | Alpha I B.1.17(U.K.) |
| B.1.1.33.etc | C.1.1.etc. | |
Certificate






