COVID-19 Antigen Rapid Test (Swab), Nasopharyngeal Swab/Nasal Swab,
SARS-CoV-2 Antigen
| Product features | Parameters |
| Principle | Chromatographic Immunoassay |
| Format | Cassette |
| Specimen | Oral fluid/Nasopharyngeal Swab/Nasal Swab |
| Certificate | CE |
| Reading Time | 15 minutes |
| Pack | 20 T |
| Storage Temperature | 2-30°C |
| Shelf Life | 2 Years |
COVID-19 Antigen Rapid Test is a rapid chromatographic immunoassay
for the qualitative detection of SARS-CoV-2 Nucleocapsid Protein antigens
in swab specimen. For professional in vitro diagnostic use only.
INTENDED USE
The COVID-19 Antigen Rapid Test (Swab) is a rapid chromatographic
immunoassay for the qualitative detection of SARS-CoV-2 Nucleocapsid Protein
antigens in swab specimens from individuals with suspected SARS-CoV-2 infection in
conjunction with
clinical presentation and the results of other laboratory tests.
Results are for the detection of SARS-CoV-2 Antigens. An antigen is
generally detectable in upper respiratory specimens during the acute phase of
infection. Positive results indicate the presence of viral antigens, but clinical
correlation with patient history
and other diagnostic information is necessary to determine
infection status. Positive results do not rule out bacterial infection or co-infection with
other viruses. The agent detected may not be the definite cause of disease.
Negative results do not preclude SARS-CoV-2 infection and should
not be used as the sole basis for treatment or patient management decisions. Negative
results should be treated as presumptive and confirmed with a molecular assay, if
necessary for patient management. Negative results should be considered in the context of
a patient’s recent exposures, history and the presence of clinical signs and symptoms
consistent with COVID-19.
SUMMARY
The novel coronaviruses belong to the β genus. COVID-19 is an acute
respiratory infectious disease. People are generally susceptible. Currently,
the patients infected by the novel coronavirus are the main source of infection;
asymptomatic infected people can also be an infectious source. Based on the current
epidemiological investigation, the incubation period is 1 to 14 days, mostly 3 to 7 days. The main
manifestations include fever, fatigue and dry cough. Nasal congestion, runny nose,
sore throat, myalgia and diarrhea are found in a few cases.
PRINCIPLE
The COVID-19 Antigen Rapid Test (Swab) is a qualitative
membrane-based immunoassay for the detection of SARS-CoV-2 Antigens in human swab
specimen. SARS-CoV-2 antibody is coated in the test line region. During
testing, the specimen reacts with SARS-CoV-2 antibody-coated particles in the test. The
mixture then migrates upward on the membrane by capillary action and reacts with
the SARS-CoV-2 antibody in test line region. If the specimen contains SARS-CoV-2
Antigens, a colored line will appear in test line region as a result of this. If the
specimen does not contain antigens to SARS-CoV-2, no colored line will appear in the test
line region, indicating a negative result. To serve as a procedural control, a colored line
will always appear in
the control line region, indicating that the proper volume of
specimen has been added and membrane wicking has occurred.
STORAGE AND STABILITY
Store as packaged in the sealed pouch at room temperature or
refrigerated (2-30°C). The test is stable through the expiration date printed on the
sealed pouch. The test must remain in the sealed pouch until use. DO NOT FREEZE. Do not
use beyond the expiration date.
SPECIMEN COLLECTION, TRANSPORT AND STORAGE
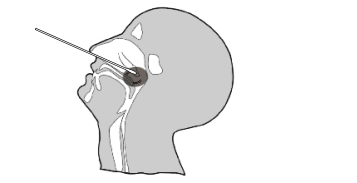
Nasopharyngeal Swab Specimen Collection
1. Insert a sterile swab into the nostril of the patient, reaching
the surface of the posterior nasopharynx.
2. Swab over the surface of the posterior nasopharynx.
3. Withdraw the sterile swab from the nasal cavity.
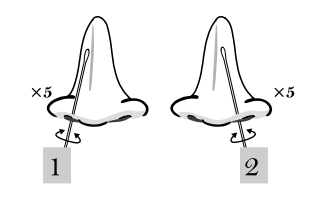
Nasal Swab Specimen Collection
1. Insert a sterile swab less than one inch (about 2 cm) into a
nostril (until resistance is met at the turbinates).
2. Rotate the swab 5-10 times against the nasal wall. Using the
same swab repeat the collection procedure with the second nostril.
3. Withdraw the sterile swab, avoid excess volume and high-viscous
nasal discharge.
Caution:
If the swab stick breaks during specimen collection, repeat
specimen collection with a new swab.
Specimen transport and storage
Specimens should be tested as soon as possible after collection. If
swabs are not been processed immediately, it is highly recommended the swab sample is
placed into a dry, sterile, and tightly sealed plastic tube for storage. The swab
specimen in dry and sterile condition is stable for up to 24 hours at 2-8°C.
SPECIMEN PREPARATION
Only the extraction buffer and tubes provided in the kit is to be
used for swab specimen preparation.
Please refer to the Procedure card for detailed information of
Specimen Extraction.
1.Place the swab specimen in the Extraction tube with Extraction
Buffer. Rotate the swab for approximately 10 seconds while pressing the head against
the inside of the tube to release the antigen in the swab.
2. Remove the swab while squeezing the swab head against the inside
of the Extraction tube as you remove it to expel as much liquid as possible from the
swab. Discard the swab in accordance with your biohazard waste disposal protocol.
*NOTE: The storage of the specimen after extraction is stable for 2
hours at room temperature or 24 hours at 2-8°C.
INTERPRETATION OF RESULTS
(Please refer to the illustration above)
POSITIVE:* Two distinct colored lines appear. One colored line
should be in the control region (C) and another colored line should be in the Test
region (T). Positive result in the Test region indicates detection of SARS-CoV-2
antigens in the sample.
*NOTE: The intensity of the color in the test line region (T) will
vary based on the amount of SARS-CoV-2 antigen present in the sample. So any shade of
color in the test region (T) should be considered positive.
NEGATIVE: One colored line appears in the control region (C). No
apparent colored line appears in the test line region (T).
INVALID: Control line fails to appear. Insufficient specimen volume
or incorrect procedural techniques are the most likely reasons for control line
failure. Review the procedure and repeat the test with a new test. If the problem
persists, discontinue using the test kit immediately and contact your local distributor.
Order Information
| Cat. No. | Product | Specimen | Pack |
| ICOV-502 | COVID-19 Antigen Rapid Test | Nasopharyngeal Swab | 25T |
| INCP-502 | SARS-CoV-2 Antigen Rapid Test | Nasopharyngeal Swab/Nasal Swab | 20T |
| ICOV-802 | COVID-19 Antigen Rapid Test | Oral Fluid | 20T |
| INCP-502-N | COVID-19 Antigen Rapid Test | Nasal Swab | 20T |

