This product uses cross-current immunoassay to be used for
qualitative testing of the novel coronavirus antigen in
nasopharyngeal and oral pharyngeal swabs of suspected patients.
Wide range of applications: suitable for hospitals, disease control
centers, communities, airports, train stations, customs, schools,
businesses, etc..
【Sample Requirements】
We recommend aseptic sampling using a polyester sponge with a PP
(polypropylene) stick.
(1) How to grate the oropharyngeal specimen: The subject tilts his
head slightly and opens his mouth to expose the amygdala of the
pharynx on both sides. Wipe both sides of the pharyngeal amygdala
at least 3 times through the root of the tongue. Then wipe above
and below the posterior wall of the pharynx at least three times.
(2) How to grate the nasopharyngeal sample: The grabber lightly
supports the grabbed person's head with one hand, holds the wipe
with one hand, inserts it into the nostril, and slowly back along
the bottom of the inferior nasal passage. Infiltrate. The nasal
meatus is bent, so use excessive force to avoid traumatic bleeding.
When the tip reaches the posterior wall of the nasopharynx, gently
turn it once (stop for a while if you have a reflex cough) and
slowly remove it.
(3) Sample processing: The swallowed sample must be treated with
the sample buffer provided by this kit as soon as possible (if not
treated immediately, it must be dried, sterilized and stored in a
sealed plastic tube immediately. Do not), and store at 2 ° C-8 ° C
for no more than 24 hours; store at -70 ° C for extended periods of
time, but avoid repeated freeze-thaw.
Test Method
Bring all reagents to room temperature before testing. The test
should be conducted at room temperature.
I. Sample extraction (see Figure 1)
1. Put 400 μL (about 10 drops) of buffer solution vertically in the
sample extraction tube, insert it into the solution, bring it close
to the inner wall and rotate it about 10 times to dissolve as much
as possible.
2. Squeeze the tip of the sample along the inner wall of the
extraction tube, leave as much liquid as possible in the tube, and
take out the sample.
3. Cover the transmitter.

II. Test Procedure (See Figure 2)
1. Remove the test card from the sticker bag.
2. Add 2 drops (about 80 μl) of the treated sample extract to the
addition hole of the test card and activate the timer.
3. Place the test card at room temperature for 15 minutes before
reading the results. After 20 minutes, the result was invalid.

Interpretation of test results
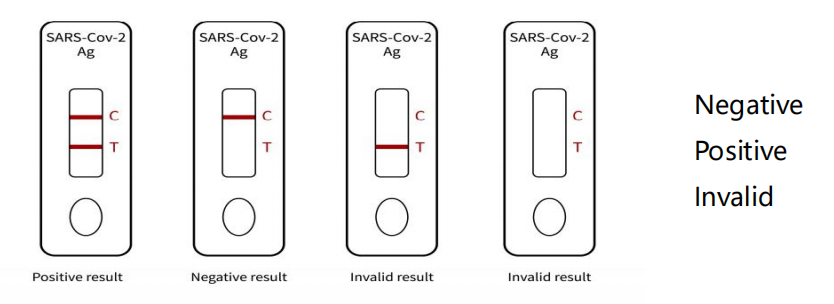
(1) Invalid result: The test was invalid because the reaction line
did not appear on the quality control line (C line). This
experiment should be redone.
② Negative result: Red ribbon, color of quality control line (C
line).
③ Positive result: Two red bands, test line (T line) and quality
control line (C line) are both displayed in color.
Company profile
Zhongxiu Science And Technology Co.,Ltd., is a high-tech enterprise
engaged in the research and development, production and operation
of in vitro diagnostic products. The in vitro diagnostic products
developed by the company cover POCT series, microbial series,
biochemical series and immune series reagents and supporting
instruments.
The company has always adhered to the core concept of "fast and
accurate, living up to life", committed to providing society with
excellent products and services, and contributing to the cause of
human health.
Zhongxiu Qualifications
DOC







