Henan Lantian Medical Supplies Co.,Ltd. |
|
The kit will be used to qualitatively test for new corona virus
antigens in 2019. Collect human saliva/sputum samples.
The new corona virus is a new corona virus that may cause viral
pneumonia. Clinical symptoms include fever, tiredness, dry cough,
stuffy nose, runny nose, sore throat, and diarrhea. Severe patients
usually suffer from dyspnea and/or hypoxemia after one week, and
severe patients suffer from acute dyspnea syndrome, septic shock,
metabolic ados, and coagulation dysfunction.
The legitimacy of new coronavirus antigens in suspected
saliva/sputum samples was detected by Imuno Cromastersic. Antigens
are usually detected in saliva/sputum samples of infection during
the acute period. A positive result indicates the presence of viral
antigens, but clinical correlations between the history and other
diagnostic information are also necessary to determine the status
of infection. A positive result does not rule out bacterial or
other virus-related infections. The pathogen detected may not be
the correct cause of infection. The results of negative illness do
not exclude corona viruses newly infected in 2019 and cannot be
used as a basis for treatment or patient management decisions,
including infection management decisions. A negative result should
be confirmed by nucleic acid analysis based on the patient's recent
exposure, past medical history, clinical symptoms, and new
coronavirus-related signs as needed.
The kit consists of test card, sample buffer storage and cotton
stick.
Test card: aluminum foil bag, desiccant, test paper, plastic card
composition. The paper consists of suction paper, nitrocellulose
mixture, sample pad, rubber pad and rubber film. 2019
Nitrocellulose membrane T-line (test line) winding - NCOV Ab,
C-line (QC line) winding Goat anti-rat polyclonal antibody, binding
pad includes 2019-NCOVAB label.
Sample buffer: phosphoric acid, nitrile azo, etc.
The temperature is maintained at 2-30 ℃ for 18 months.
The aluminum foil package will remain valid for 1 hour after
opening.
Production Lot Number: Refer to the label for details.
Expiry date: Refer to the label.
(1) Saliva sampling: do not eat chewing gum or cigarette products
within 30 minutes after saliva sampling. Place your tongue on the
mouth lid to collect the roots of saliva. Place a disposable
disinfecting swab under the saliva sample. The tip of the tongue
should be applied for 10 seconds or more, fully immersed in saliva,
and rotated 5 times or more. (See Table 1)
Note: Saliva sample is not collected correctly.
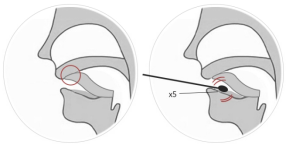
Figure 1 Methodsfor saliva sample collection

Figure 2 Methods for sputum sample collection
(2) Sputum collection: rinse the sputum with clean water, cough
continuously, put the sputum into the test bag, and wipe the sample
with a disposable germicidal cotton stick. (Refer to Figure 2)
(3) Sample processing: Collected samples are processed by the
sample buffer memory provided by the supporting component (samples
not immediately processed must be stored in a dry and germicidal
sealed container). 2-8 degrees Celsius to 24 hours, longer than 70
degrees Celsius (however, please avoid repeated freezing and
thawing).
Please read the instructions carefully before the exam. All
reagents will be returned to room temperature for testing.
Processing example (see Figure 3).
(1) Insert the sample exchanger into the sample buffer memory,
rotate it around the inner wall for about 10 times, and dissolve
the sample in solution as far as possible.
(2) Press down the casing head along the inner wall, the liquid
flows into the pipe, and the casing is removed and discarded.
(3) Cover with small water drops.

Figure 3 Sample processing

Figure 4 Test procedure
2. The test process (see Figure 4).
(1) Remove the test card.
(2) Add 2 drops (about 80L) of the processed sample extract into
the sample well of the test card and start timing.
(3) Read the card for 15 minutes at room temperature, and the
result is invalid after 20 minutes.

Figure 5 Interpretation of test results
1. This product is only used for qualitative testing as an aid to
INVitro diagnosis.
2. This product is suitable for saliva and sputum samples. Other
types of samples may be incorrect or invalid.
3. Patients who do not have sputum samples should be examined using
nasopharyngeal swans.
4. Please add a suitable amount of samples for testing. Or, if
there are too many samples, the results may be incorrect.
5. The test results of the reagent are for clinical reference only,
and should not be used as the sole basis for clinical diagnosis and
treatment. The diagnosis was made after a comprehensive evaluation
of all clinical and examination results.
1. Detection limits: Inactive SARS-COV-2 virus cultures were used
in this study. The detection limit was 610TCID50 / mL.
2. The test must use the company's standard materials, and the test
results must meet the requirements of the company's standard
materials.
2.1 Qualification rate of positive control: P1-P5 of positive
control of enterprises.
2.2 Pass rate of negative control: N1-N10 negative control test is
negative.
2.3 Test limit: please refer to L1-L3. L1 is negative, L2 and L3
are positive.
2.4 Reproducibility: J1 and J2 were 10 times positive.
3. Cross-reactivity: Inject samples with the following
microorganisms and viruses at specified concentrations to assess
potential interference with the new coronavirus antigen testing
program in 2019.
4. Interference: To evaluate the potential interference in the
2019-NCOV Ag detection step, add the following agents at the
specified concentration. The results showed that all kinds of drugs
did not interfere with the results of the reagent.
5. Hook effect: There is no hook effect within the high
concentration range of 1.0106 TCID50/mL.
6. Clinical trial: Saliva and sputum samples were detected
separately using RT-PPCR sputum reagent as a comparative reagent.
120 positive and negative samples were selected for each sample
size (RT-PPCR test), and the second test was performed using XIUS
reagent. The research results are summarized as follows.
1. This product is used for INVitro diagnosis.
This product is disposable and can't be reused.
3. Please strictly follow the instructions of the reagent and read
the instructions carefully before the experiment.
4. Do not perform the test under severe environmental conditions
(including 84 disinfectant, dust, sodium hypochlorite, acid and
alkali, acetaldehyde and other corrosive gases with high
concentration). Laboratory disinfection needs to be carried out
after the experiment.



