The product adopts cross flow immunoassay for qualitative detection
New Coronavirus antigen in nasopharyngeal and oropharyngeal swabs
of suspected patients
Wide application: applicable to hospitals, CDC, community, airport,
station, customs, schools, enterprises, etc.
It is recommended to use a polyester sponge sample with a PP
(polypropylene) rod for sterile sample collection.
(1) Collection method of oropharyngeal specimens: the collector's
head is slightly tilted, the mouth is open, and the pharyngeal
tonsils on both sides are exposed. Apply the swab across the base
of the tongue from the base of the tongue to the bilateral
pharyngeal tonsils. The recipient should wipe the posterior
pharyngeal wall up and down at least 3 times with a cotton swab.
(2) Nasopharynx sample collection method: the sampler gently holds
the head of the person to be collected with one hand, holds the
swab in one hand, inserts the swab into the nostril, and slowly
penetrates backward along the bottom of the inferior meatus. As the
nasal meatus is curved, do not use excessive force to avoid
traumatic bleeding. When the top of the swab reaches the posterior
wall of the nasopharynx, gently rotate the swab once (pause for
reflex cough), and then slowly remove the swab
(3) Sample processing: the sample collected shall be treated with
the sample buffer solution provided by this reagent box as soon as
possible (if not treated immediately, the sample shall be
immediately stored in a dry, sterilized and sealed plastic pipe),
and shall not exceed 24h at 2 ℃ to 8 ℃- The storage at 70 ℃ is
long-term, but repeated freezing and thawing should be avoided.
Test Method
Restore all reagents to room temperature before testing. The test
should be conducted at room temperature.
I. Extraction of specimens (see Figure 1)
1. Vertically add 400μL (approximately 10 drops) of sample buffer
into the sample extraction tube, then insert the sampled test piece
into the solution in the sample extraction tube, and rotate it
against the inner wall about 10 times to make the sample as
dissolved in the solution as possible.
2. Squeeze the cotton swab head of the test piece along the inner
wall of the extraction tube to keep the liquid in the tube as much
as possible, take out and discard the test piece.
3. Cover the dripper.

II. Test Procedure (See Figure 2)
1. Take out the test card from the sealed bag.
2. Add 2 drops (about 80μL) of the processed sample extract to the
sample hole of the test card, and then start the timer.
3. Read the result when the test card is placed at room temperature
for 15 minutes. The reading result is invalid after 20 minutes.

Interpretation of test results
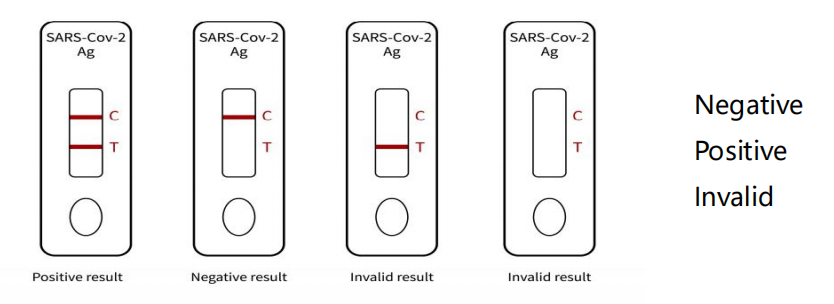
Diagram of test card result judgment:
① Invalid results: there was no reaction line in the quality
control line (C line), and the test was invalid. The experiment
should be redone
② Negative results: red ribbon, color of quality control line (C
line)
③ Positive results: two red bands, test line (t line) and quality
control line (C line) were displayed in color.







