HIV(1+2) Antibody Rapid Test Kit
For qualitative detection of HIV(1+2) Antibodies in serum/plasma
and whole Blood
Main Features
► Sensitivity: 100%
► Specificity: Higher than 99%
► Simple: No Instrument Required
► Ambient Storage
► Reliable: able to differentiate HIV Type I and Type II
► Certified by Authoritative Certification
► Unique 3-line Patented Design
► Winner of "the 2008 National HIV Antigen Diagnostic Kit for
Clinical Performance Assessment"
Intended Use
The HIV-1/2 Rapid Diagnostic Test (RDT) is a qualitative test for
the detection of antibodies to Human Immunodeficiency Virus type 1
and 2 (HIV-1/2) in human serum/plasma. It is considered as an
initial screening test for HIV-1/2 antibodies. All positive
specimens must be confirmed with Western Blot or other qualified
EIA.

Principle
HIV-1/2 RDT is a chromatographic immunoassay (CIA) for the
detection of antibodies to HIV-1/2 in human serum/plasma. HIV-1/2
specific antigens are precoated onto membrane as a capture reagent
on the test region. During the test, specimen is allowed to react
with the colloidal gold particles, which have been labeled with
HIV-1/2 specific antigens. Antibodies to HIV-1/2, if present, a red
colored band will develop on the membrane in proportion to the
amount of HIV-1/2 antibodies present in the specimen. Absence of
this red colored band in the test region suggests a negative
result. To serve as a procedural control, red colored band in the
control region will always appear regardless the presence of
antibodies to HIV-1/2.
Assay Procedure
Serum/Plasma
Add 70-100 ul or 2-3 drops of serum or plasma into sample well.
Observe the result in 5 –30 minutes
Whole Blood
Add 1 drop of whole blood into sample well, after all blood
completely absorbed, add 1 drop of whole blood diluent. Observe the
result in 5-30 minutes.
Interpretation of Results
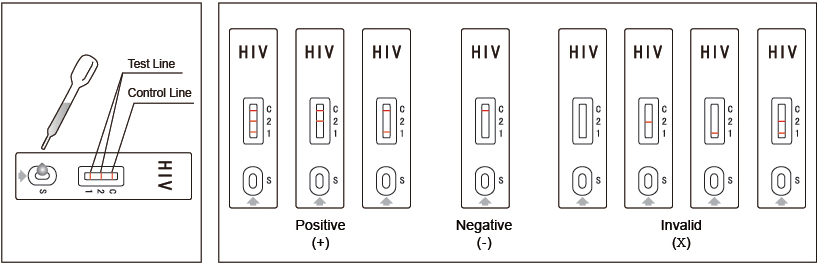
Negative:
No apparent band in the test region (1 and 2), only one red band
appears in the control region (C). This indicates that no HIV1/2
antibodies have been detected.
Positive:
In addition to the band in the control region (C), other one or two
red bands will appear in the test region (1 and 2). This indicates
that the specimen contains HIV1/2 antibodies.
Invalid:
If no band appears in the control region(C), regardless of the
presence or absence of line in the test region (1 and 2). It
indicates a possible error in performing the test. The test should
be repeated using a new device.

Storage
Store the test kits at temperature 4-25°C,in the sealed pouch for
the duration of the shelf life (24 months).
Reagents and Materials Provided
Each kit contains:
1. 40 test cassettes(individually pouched)
2. Each pouch contains one cassette with one desiccant bag
3. One bottle of diluent buffer(5ml)
4. 40 disposable plastic droppers
5. Instruction for use
Materials Required But Not Provided
1. Timer or stopwatch
2. Blood collection devices, for the testing of venous whole blood,
serum or plasma
3. Biohazard disposal container
4. Disposable gloves
For finger stick samples, the following materials are required:
Alcohol pad
Sterile lancet
Sterile gauze or cotton
Warning
For Invitro Diagnostic Use ONLY
Read the package insert completely before use. It is very important
that the correct procedure is followed. Fail to add the patient
sample may lead to a false negative result (i.e.a missed positive)
.






