Jinan Fosychan International Trading Co., Ltd. |
|
Verified Suppliers
|
|
2ml Injection Deep Non Cross Linked Hyaluronic Acid Filler For Joint Injection
Our Company
Business Type: Trading Company
Main Products: Fosyderm cross-linked ha
Main Markets: Mid East, South America, North America, Eastern
Europe.
Location: Shan dong, China (Mainland)
Total Employees: 51 - 100 People
Year Established: 2017



Fosyderm non cross-linked hyaluronic acid may be recommended forosteoarthritis of the knee joint, shoulder, elbow or ankle.Hyaluronic acid helps to lubricate the joint and may be a treatment option if the surfaces of the joint have become rough, but there's not too much inflammation.
Sodium Hyaluronate
Which is a polymer polysaccharides of biological materials formed
by N-acetyl glucosamine and glucuronic acid through iterative
connected, has a high degree of viscoelasticity, plasticity,
permeability and good biocompatibility. This product is
manufactured with raw material of comb based on the process of fine
biochemical technology, separation, purification and refining. The
gel contains 1% HMW of sodium hyaluronate and physiological buffer.
This product, which is a colorless, odorless, sterile, pyrogen free
transparent viscous liquid, adopts the aseptic processing
technology to filter sterilization
Function
This product is composed by high purified hyaluronic sodium and
physiological buffer ,and is of colorless and transparent
gelatinous solution Hyaluronic sodium can stimulate its own
synovial memberance to produce sodium hyaluronate of high molecular
weight,which helps alleviate arthralgia,increase the range of
mobility,eliminate synovial inflammation and delay the development
of the disease.
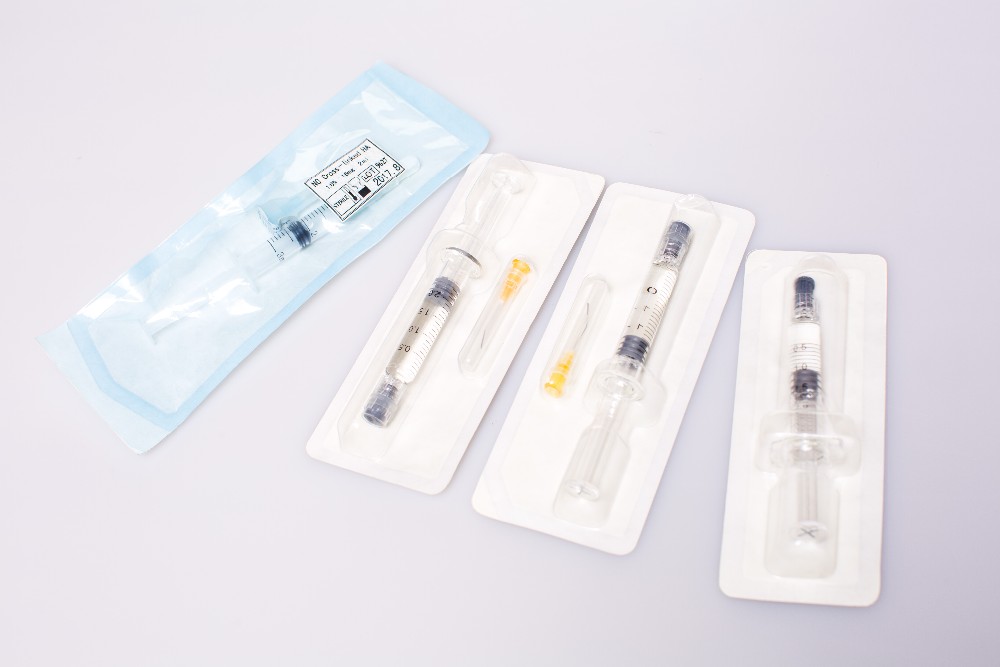
| Name | Fosyderm 3ml Non Cross-Linked Injectable HA Dermal Fillers |
| Packaging | 1ml/syringe;2ml/syringe;3ml/syringe |
| Concetration | 20mg/ml |
| Storage | Store between 2℃ and 20℃,Do not Freeze. Relative humidity no more than 80%, well ventilated and clean, no corrosive gas, protect from light. Do not uses after expiry date |
1. Constitute :
This product is composed by high purified hyaluronic sodium and
physiogical buffer ,and is of colorless and transparent gelatinous
solution Hyaluronic sodium can stimulate its own synovial
memberance to produce sodium hyaluronate of high molecular
weight,which helps alleviate arthralgia,increase the range of
mobility,eliminate synovial inflammation and delay the development
of the disease.
2.Character:
High-purity ,high viscoelastic,high transparent can maintain a
clear surgical environment ,it is a perfect viscoelastic protective
agent to assist the orthopedics surgery
3.Overview:
Its macromolecule and good viscoelasticity help lubricate,cover the
barrier,buffer stress and inhibit the inflammatory response in the
articular cavity.And it can stimulate its own synovial memberance
to produce sodium hyaluronate of high molecular weight,which helps
alleviate arthralgia,increase the range of mobility,eliminate
synovial inflammation and delay the development of the disease.
4.Packing:(BD)syringe needle or as requested
1ml/Syringe,2ml/syringe,3ml/syringe

| Type | Non cross link ha knee joint dermal filler |
| Composition | 20mg/ml |
| Specification | 1ml 2ml 3ml |
| Particle size | 0.10~0.15 |
| Approximately number of gel(ml) | 100,000 |
| Storage | Room temperature |
| Duration | 6~12months |
| Function | Lubrication |
Our advantages:
1) non-animal origin.
2) long duration.
3) BD syringe
4) biodegradable.
5) Non cross-linking
Why choose us?
1. Factory price
2. Minimum order quantity (acceptable within 10 PCS)
3. OEM acceptance
4. Raw material safety is 100%.
5. Rapid transportation
6. 8/24 hours service.
7. 100% security payment.
8. The syringe needle is made by BD company, strict quality, high
quality and safety!

Hyaluronic Acid Injection/Filler Injection
it is non-animal ingredient cross-linked sodium hyaluronate gel for
injection.Soft tissue