Hebei R&M Healthcare Company Ltd. |
|
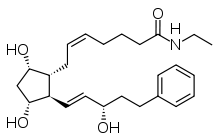 | |
| Clinical data | |
|---|---|
| Trade names | Lumigan |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a602030 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | Topical (eye drops) |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | Low |
| Protein binding | 88% |
| Onset of action | 4 hrs |
| Elimination half-life | 45 min after IV application |
| Duration of action | ≥ 24 hrs |
| Excretion | 67% renal, 25% fecal |
| Identifiers | |
IUPAC name
| |
| CAS Number |
|
| PubChem CID |
|
| IUPHAR/BPS |
|
| DrugBank |
|
| ChemSpider |
|
| UNII |
|
| KEGG |
|
| ChEBI | |
| ChEMBL | |
| ECHA InfoCard | 100.170.712 |
| Chemical and physical data | |
| Formula | C25H37NO4 |
| Molar mass | 415.566 g/mol |
| 3D model (JSmol) | |
Bimatoprost (marketed in the US, Canada and Europe by Allergan, under the trade name Lumigan) is a prostaglandin analog used topically (as eye drops) to control the progression of glaucoma and in the management of ocular hypertension. It reduces intraocular pressure (IOP) by increasing the outflow of aqueous fluid from the eyes. In December 2008, the indication to lengthen eyelashes was approved by the U.S. Food and Drug Administration (FDA); the cosmetic formulation of bimatoprost is sold as Latisse /ləˈtiːs/.