Rapid Test Kit SARS-CoV-2 Antigen & Influenza A/B Rapid Test
Kit Professional Detection Rapid Test Kit
Intended Use
The SARS-CoV-2 Antigen & Influenza A/B Rapid Test Kit
(Immunochromatography) is a lateral flow chromatography immunoassay
intended for the qualitative detection and differentiation of
nucleocapsid protein antigens from SARS-CoV-2, influenza type A and
influenza type B in the same time directly from
nasal/nasopharyngeal (NS/NP) swab, oropharyngeal (OP) swab and
saliva specimens from patients with signs and symptoms of
respiratory viral infection. It is for professional use
only. The kit is intended for use by trained clinical
laboratory personnel and individuals trained in point of care
settings. For prescription use only. For in vitro diagnostic use
only.
Specification
| Item | Antigen test strip performance against PCR | Influenza A test strip performance against PCR | Influenza B test strip performance against PCR |
| Sensitivity | 98.03% | 93.30% | 97.00% |
| Specificity | 100.00% | 91.00% | 96.40% |
Main Components
- 20 Test cassettes
- 2 Sample extraction Buffer
- 20 Sample tubes
- 20 Swabs
- 1 Tube Stand
- 1 Instruction Manual

Feature
- High Specificity and Sensitivity
- Safety and Reliability, individual package keep clean
- Simple operation step, one step to do the test
- Fast reacton read the result within15 min

Use Step
- Take out the test cassette from the sealed pouch, place it on a
clean and level surface with the sample port well up.
Apply 2 full drops of the treated sample (60μl-70μl) vertically
into each of the two sample wells of the test cassette.
Observe the test results immediately within 15~20 minutes, the
result is invalid over 20 minutes.

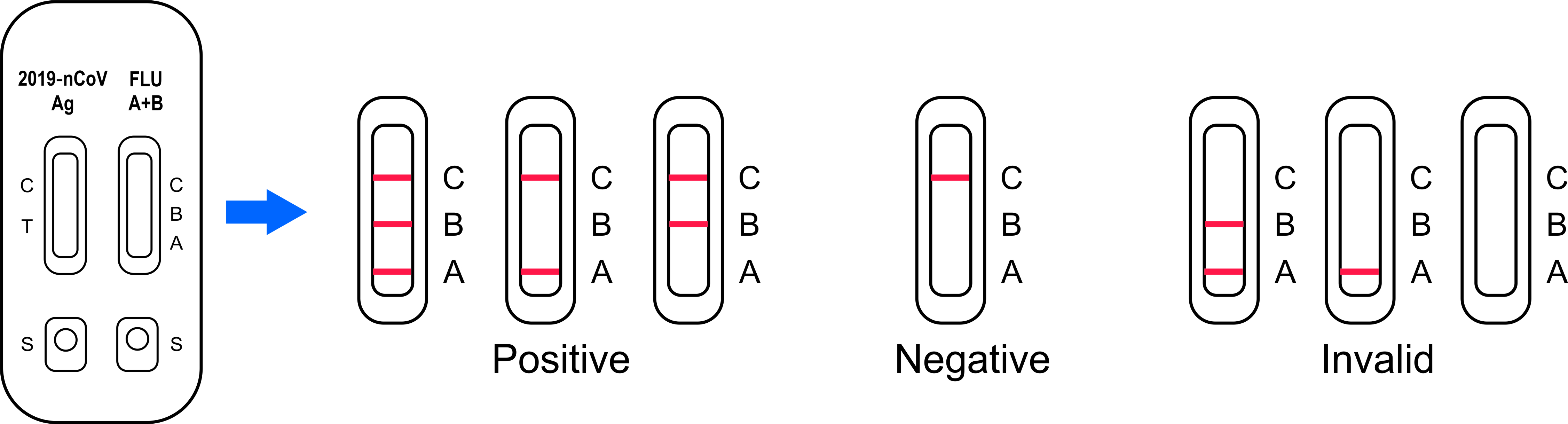
Interpretation Of Result
POSITIVE: The presence of T (T on nCoV /A or B on Flu) and C lines within
the reaction window indicate a positive result on SARS-CoV-2 or Flu
A and/or B infection or co-infection.
NEGATIVE: One colored line appears in the control region(C). No apparent
colored line appears in the test region (T on nCoV /A or B on Flu).
The negative result does not indicate the absence of analytes in
the sample, it only indicates the level of tested analytes in the
sample is less than the minimum detection limit
INVALID: No colored lines appear, or control line fails to appear,
indicating that the operator error or reagent failure. Verify the
test procedure and repeat the test with a new testing device.
PRINCIPLE
- The SARS-CoV-2 Antigen & Influenza A/B Rapid Test Kit employs a
lateral flow chromatographic technology to detect the presence of
the nucleocapsid protein antigen directly from influenza A,
influenza B, and SARS-CoV-2.
- After the patient sample is collected and treated with the antigen
extraction buffer, the viral nucleoproteins antigen will be
exposed.
- Add the extracted specimen into the test cassette, the specimen
will migrate forward along with the test strips through capillary
effect.
- If Influenza A, Influenza B, SARS-CoV or SARS-CoV-2 viral antigen
is present, they will be captured and detected on the T or A/B
line, respectively on each test strips within 15 minutes from the
addition of the samples, resulting in purplish red band on the test
region, indicating a positive result.
- If the nucleocapsid protein antigen is not present or present at
very low levels in the sample, there is no red line appears in “T”
or "A/B" positions. The “Control Line” (C) is used for procedural
control.
- Control line should always appear if the test procedure is
performed properly and the test reagents of control line are
working.
Other Information
- This reagent is a qualitative detection reagent, which cannot
determine the exact content of antigen.
- The test results of this reagent are only for the reference of
clinicians and should not be taken as the sole basis for clinical
diagnosis and treatment. Clinical management of patients should be
considered in the light of their symptoms/signs, medical history,
other laboratory tests and treatment responses.
- Restricted by antigen detection reagent method, the lowest
detection limit (sensitivity analysis) is generally lower than that
of nucleic acid detection, so the researchers deal with negative
result to give more attention, should be combined with other test
results comprehensive judgment, advice to doubt the negative result
of nucleic acid detection or virus isolation culture identification
method for review.



