Labnovation Technologies, Inc. |
|
Rapid Test Kit Antigen & Influenza Rapid Test SARS-CoV-2
Antigen And Influenza A/B Rapid Test Kit
Intended Use
The rapid Test Kit is intended for the qualitative detection and
differentiation of nucleocapsid protein antigens from SARS-CoV-2,
influenza type A and influenza type B in the same time directly
from nasal/nasopharyngeal (NS/NP) swab, oropharyngeal (OP) swab and
saliva specimens from patients with signs and symptoms of
respiratory viral infection. It is for professional use only. It is
an aid in the diagnosis of the patients with suspected SARS-CoV-2
or influenza infection in conjunction with clinical presentation
and results of other laboratory tests. Results from this test kit
should not be used as the sole basis for diagnosis.
Specification
| Item | Antigen test strip performance | Influenza A test strip performance against PCR | Influenza B test strip performance against PCR |
| Sensitivity | 98.03% | 93.30% | 97.00% |
| Specificity | 100.00% | 91.00% | 96.40% |
Main Components

Feature

Sample Collection
Sample Treatment
Add 550μl sample extraction(about 22-24 drops) into the sample
tube, dip the swab after collecting the sample into the sample
extraction liquid, fully immerse the tip of the swab, rotate and
squeeze the swab 10 times, then pull out the swab, and take the
stranded liquid as the sample to test cassette.
Use Step
Apply 2 full drops of the treated sample (60μl-70μl) vertically into each of the two sample wells of the test cassette.
Observe the test results immediately within 15~20 minutes, the result is invalid over 20 minutes.

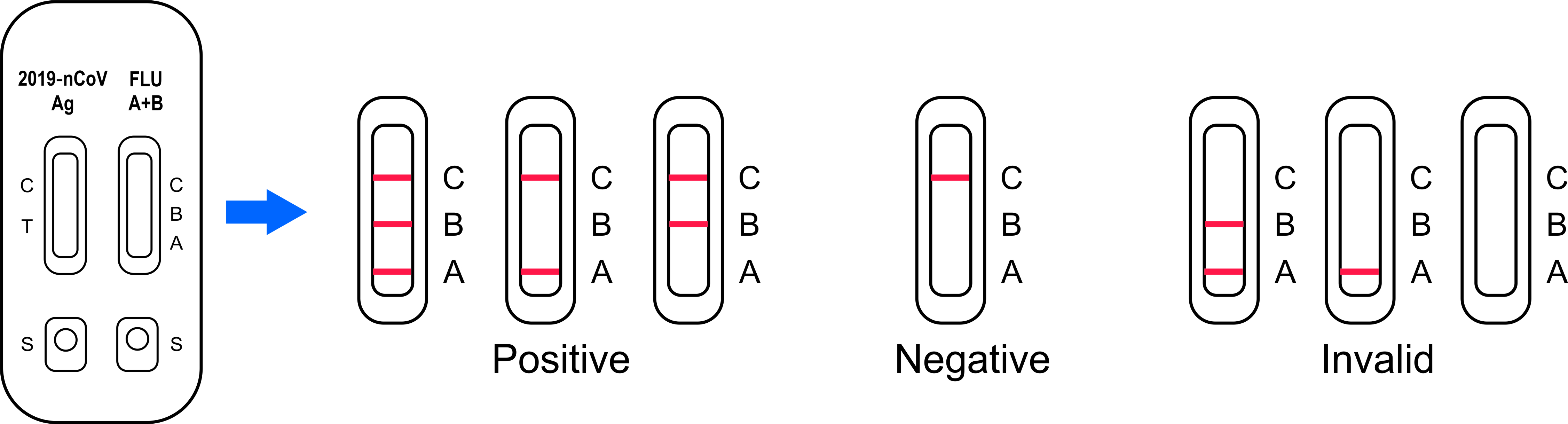
Interpretation Of Result
POSITIVE: The presence of T (T on nCoV /A or B on Flu) and C lines within the reaction window indicate a positive result on SARS-CoV-2 or Flu A and/or B infection or co-infection.
NEGATIVE: One colored line appears in the control region(C). No apparent colored line appears in the test region (T on nCoV /A or B on Flu). The negative result does not indicate the absence of analytes in the sample, it only indicates the level of tested analytes in the sample is less than the minimum detection limit
INVALID: No colored lines appear, or control line fails to appear, indicating that the operator error or reagent failure. Verify the test procedure and repeat the test with a new testing device.
Other Information