Human Immunodeficiency Virus HIV 1/2 and Syphilis Treponema
Pallidum Tp Duo Rapid Test Whole Blood
INTENDED USE
The HIV 1/2 & Syphilis(TP) Rapid Test is a rapid visual
immunoassay for the qualitative, presumptive detection of
antibodies to HIV-1/HIV-2 and antibodies to Treponema Pallidum (TP)
in human whole Blood, serum or plasma specimens. This kit is
intended for use as an aid in the diagnosis of HIV infection.
PRINCIPLE
The HIV 1/2 Rapid Test detects antibodies to HIV-1/HIV-2 and
Treponema Pallidum (TP) through visual interpretation of color
development on the internal strip.
Recombinant HIV antigens are immobilized on the test region of the
membrane. During testing, the specimen reacts with HIV antigen
conjugated to colored particles and precoated onto the sample pad
of the test. The mixture then migrates through the membrane by
capillary action and interacts with reagents on the membrane. If
there are sufficient HIV-1/HIV-2 antibodies in the specimen, a
colored band will form at the HIV test region of the membrane. The
presence of this colored band indicates a positive HIV result,
while its absence indicates a negative result.
Specific recombinant TP antigens are immobilized on the test region
of the membrane. During testing, the specimen reacts with
recombinant TP-specific antigen conjugated to colored particles and
precoated onto the sample pad of the test. The mixture then
migrates through the membrane by capillary action and interacts
with reagents on the membrane. If there are sufficient antibodies
to Treponema Pallidum (TP) in the specimen, a colored band will
form at the SYP test region of the membrane. The presence of this
colored band indicates a positive SYP result, while its absence
indicates a negative result.
The appearance of a colored band at the C control region serves as
a procedural control, indicating that the proper volume of specimen
has been added and membrane wicking has occurred.
MAIN CONTENTS
Rapid Test Cassette in Pouch
Buffer
Disposable dropper
Package insert
PRECAUTIONS
- For professional in vitro diagnostic use only. Do not use after
expiration date.
- Do not eat, drink or smoke in the area where the specimens or kits
are handled.
- Handle all specimens as if they contain infectious agents. Observe
established precautions against microbiological hazards throughout
testing and follow the standard procedures for proper disposal of
specimens.
- Wear protective clothing such as laboratory coats, disposable
gloves and eye protection when specimens are being tested.
- Humidity and temperature can adversely affect results.
- The used test should be discarded according to local regulations.
SRORAGE
Store as packaged in the sealed pouch at room temperature or
refrigerated (2-30°C).
The test is stable through the expiration date printed on the
sealed pouch.
The test must remain in the sealed pouch until use.
DO NOT FREEZE.
Do not use beyond the expiration date.
OPERATION
Bring tests, specimens, and/or controls to room temperature
(15-30°C) before use.
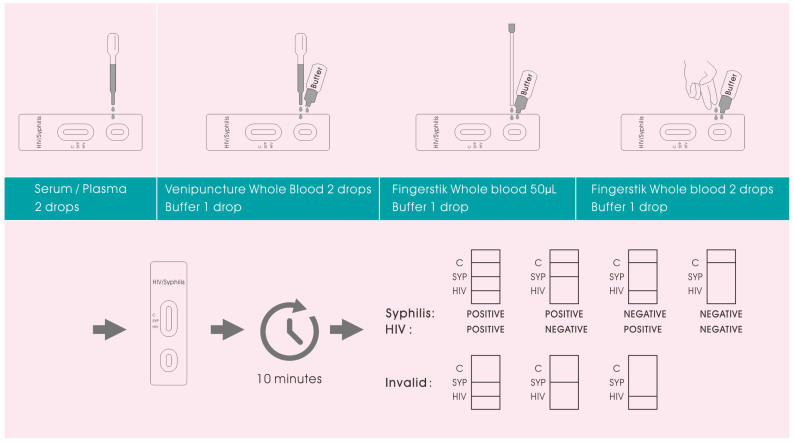
1.Remove the test from its sealed pouch, and place it on a clean,
level surface. Label the test with patient or control
identification. For best results, the assay should be performed
within one hour.
2.Using the provided disposable dropper, transfer 2 drops of
serum/plasma to specimen well (S) of the cassette, then start the
timer.
OR
Transfer 1 drop of whole blood specimen to specimen well (S) of the
cassette with the provided disposable dropper, then add 1 drop of
buffer and start the timer.
OR
Allow 2 hanging drops of fingerstick whole blood specimen to fall
into the center of specimen well (S) on the cassette, then add 1
drop of buffer and start the timer.
Avoid trapping air bubbles in the specimen well (S), and do not add
any solution to the result area.
As the test begins to work, color will migrate across the membrane.
3. Wait for the colored band(s) to appear. The result should be
read at 10 minutes. Do not interpret the result after 20 minutes.